 So addition of the electron is not possible and has positive electron gain enthalpy. The element with positive electron gain enthalpy is ……….Īrgon has completely filled configuration. Hydrogen is the first element and it has less electro negativity and down the group electro negativity decreases. Since electro negativity increases across the period from left to right. Hydrogen is the least electro negative element. Which one of the following is the least electro negative element? 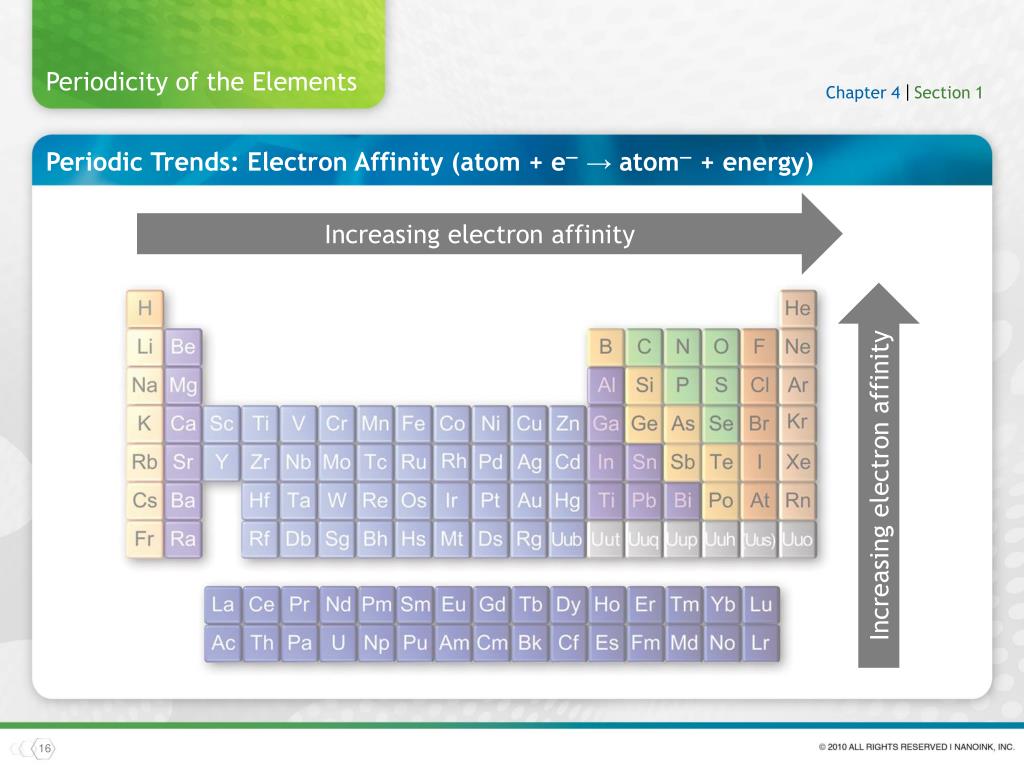
In which of the following options the order of arrangement does not agree with the variation of property indicated against it? (NEET 2016 Phase 1) The group of elements in which the differentiating electron enters the anti-penultimate shell of atoms are called – The formula of the ionic compound that can be formed between these elements is ………. The electronic configuration of the elements A and B are 1s 2, 2s 2, 2p 6, 3s 2 and 1s 2, 2s 2, 2p 5, respectively. What would be the IUPAC name for an element with atomic number 222? Tamilnadu Samacheer Kalvi 11th Chemistry Solutions Chapter 3 Periodic Classification of Elements Samacheer Kalvi 11th Chemistry Chapter 3 Periodic Classification of Elements Textual Evaluation SolvedĬhoose The Correct Answer from The Following 
ELECTRON AFFINITY DOWNLOADStudents can Download Chemistry Chapter 3 Periodic Classification of Elements Questions and Answers, Notes Pdf, Samacheer Kalvi 11th Chemistry Solutions Guide Pdf helps you to revise the complete Tamilnadu State Board New Syllabus and score more marks in your examinations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
